FRIDAY, Sept. 5, 2014 (HealthDay News) — A growing knowledge of the skin disease called psoriasis is leading to greater treatment choices, including personalized therapies, the U.S. Food and Drug Administration reports.
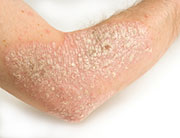
Psoriasis is an immune system disorder that causes overproduction of skin cells, resulting in scaling, pain, swelling, redness and heat. The condition affects about 7.5 million Americans.
“As we better understand the disease, researchers know more about what specific factors to target in order to develop effective treatments,” FDA dermatologist Dr. Melinda McCord said in an agency news release.
There is no cure for psoriasis, so the main goals of treatments are to stop skin cell overproduction and reduce inflammation. Current therapies include medicines applied to the skin (topical), light treatment (phototherapy), or drugs taken by mouth or given by injection.
Doctors used to take a step-by-step approach, starting patients with mild to moderate psoriasis on topical therapy. If that was ineffective, doctors moved on to phototherapy or drug treatment.
Treatment is now more patient-specific, with doctors and patients selecting a treatment based on its effectiveness, disease severity, lifestyle, risk factors and other health issues, according to the FDA.
“Tomorrow’s treatments will become even more personalized because the drugs in development now are targeting different aspects of the immune system,” McCord said.
“As we learn more about the immune pathways that lead to the development of psoriasis, we can target specific molecules for treatment and make more therapeutic options available to patients,” she explained.
Patients need to educate themselves about their condition and treatment options.
“Psoriasis has a great emotional impact on some patients. But it doesn’t have to, given the right care and treatment,” McCord said.
More information
The U.S. National Library of Medicine has more about psoriasis.
Copyright © 2024 HealthDay. All rights reserved.

