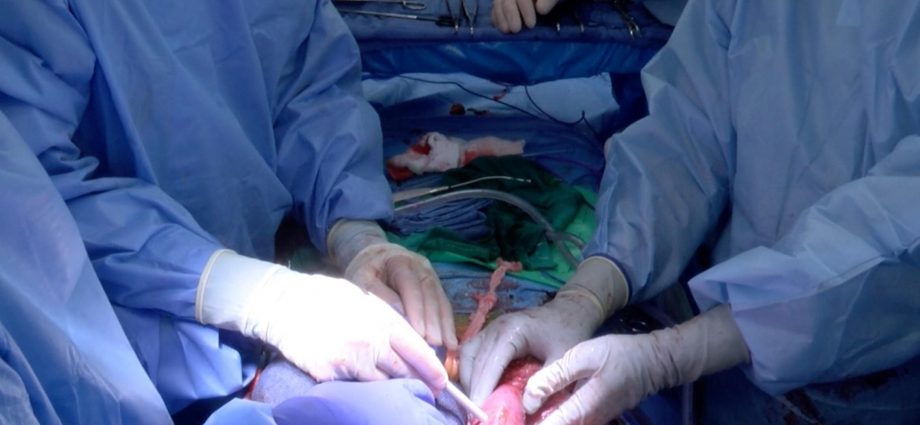
THURSDAY, Jan. 20, 2022 (HealthDay News) — In another breakthrough for animal-to-human organ transplantation, U.S. researchers say they’ve transplanted two genetically modified pig kidneys into a living human.
The recipient was Jim Parsons, 57, a brain-dead man on life support whose family agreed to allow the surgical team at the University of Alabama at Birmingham (UAB) to use his body for this research.
The kidneys functioned well and were not rejected by Parson’s body during the 77-hour period of the study.
The successful transplant comes of the heels of two similar groundbreaking transplant operations that made headlines recently: Last fall, surgeons at NYU Langone Health in New York City twice attached a pig kidney (leaving it outside the body) to a human recipient on life support; and earlier this month a team at the University of Maryland successfully transplanted a heart from a genetically modified pig into a 57-year-old man with terminal heart disease.
The new procedure is an advance on the New York City transplant in that two kidneys were used and were fully implanted into the recipient’s body in what the surgeons call a “clinical-grade” fashion, similar to what happens in human-to-human transplants.
The Birmingham team said their double-kidney transplant is yet another step forward in xenotransplantation — using organs from another species for transplant into humans. Broader success might save the lives of thousands of people who languish on donor organ waitlists every year.
“This game-changing moment in the history of medicine represents a paradigm shift and a major milestone in the field of xenotransplantation, which is arguably the best solution to the organ shortage crisis,” Dr. Jayme Locke, who directs the Comprehensive Transplant Institute at UAB’s department of surgery, said in a university news release.
‘Barriers surmounted’
The fact that the human immune system quickly recognizes and rejects foreign tissue — especially that of other species — has long been an enormous hurdle in transplant medicine.
According to the UAB team, the porcine kidneys were removed from pigs genetically modified with 10 genetic edits to reduce the risk of rejection.
The edits seemed to work: Over the course of more than three days, the transplanted kidney filtered blood and produced urine in Parson’s body, while showing no signs of rejection, the surgical team said.
The researchers said that pigs are a close fit for humans as a source for donor organs because they are easily bred and have organs of similar size.
Using a brain-dead human is what’s known as a “preclinical model” that helps doctors gain valuable insight into how xenotransplantation might work in more widespread use, the team explained.
“Our study demonstrates that major barriers to human xenotransplantation have been surmounted, identifies where new knowledge is needed to optimize xenotransplantation outcomes in humans, and lays the foundation for the establishment of a novel preclinical human model for further study,” Locke said.
According to the UAB team, over 800,000 Americans are currently living with kidney failure. Many are surviving via dialysis and may never be among the lucky ones who receive a donor organ. According to organdonor.gov, almost 107,000 Americans are on waitlists for new organs, only 39,000 transplants were performed in 2020, and 17 people die each day waiting for a transplant.
The widespread use of organs sourced from pigs could help end these tragedies.

Jim Parsons and daughter Ally. Family photo
One man’s legacy
In the study, the donor pig was first housed in a “surgically clean” facility that was rendered as free of any germs as possible, the researchers said. That was done to help minimize the threat of infection to the recipient.
When the pig kidneys were removed they were stored and transported just as human donor kidneys are.
Prior to transplant, the UAB team conducted “compatibility tests” between the patient and the gene-modified pig to see if they were a good tissue match, just as is done in human-to-human transplants. This was the first time such tests had been done between the two species, however, the researchers noted.
The anatomical placement of the pig kidneys within Parson’s body mimicked that of a real human kidney, “with the same attachments to the renal artery, renal vein and the ureter that carries urine from the kidney to the bladder,” the university said in a statement.
Standard immune-suppression drugs were used during and after the xenotransplantation, much as they would be in a human-to-human transplant.
Parsons was a registered organ donor in Alabama and had wanted his organs to help others after his death. Unfortunately, his organs weren’t suitable for transplant, the UAB team said, but Parson’s family gave permission for the xenotransplant team to use his body for the breakthrough procedure.
The surgeons removed Parson’s kidneys and then replaced them with those from the gene-modified pig.
“Mr. Parsons and his family allowed us to replicate precisely how we would perform this transplant in a living human. Their powerful contribution will save thousands of lives, and that could begin in the very near future,” Locke said. “Mr. Parsons’ gift honors his legacy and firmly establishes the viability, safety and feasibility of this preclinical model. Because of his gift, we have proposed this to be known as ‘The Parsons Model.'”
Julie O’Hara, Parson’s ex-wife and the mother of their children — Ally, David and Cole — said, “Jim would have wanted to save as many people as he could with his death, and if he knew he could potentially save thousands and thousands of people by doing this, he would have had no hesitation. Our dream is that no other person dies waiting for a kidney, and we know that Jim is very proud that his death could potentially bring so much hope to others.”
A study outlining the breakthrough procedure was published in the American Journal of Transplantation.
More information
Find out more about organ donation at the United Network for Organ Sharing (UNOS).
SOURCE: University of Alabama at Birmingham, news release, Jan. 20, 2022
Copyright © 2026 HealthDay. All rights reserved.